Papers
Johns Hopkins
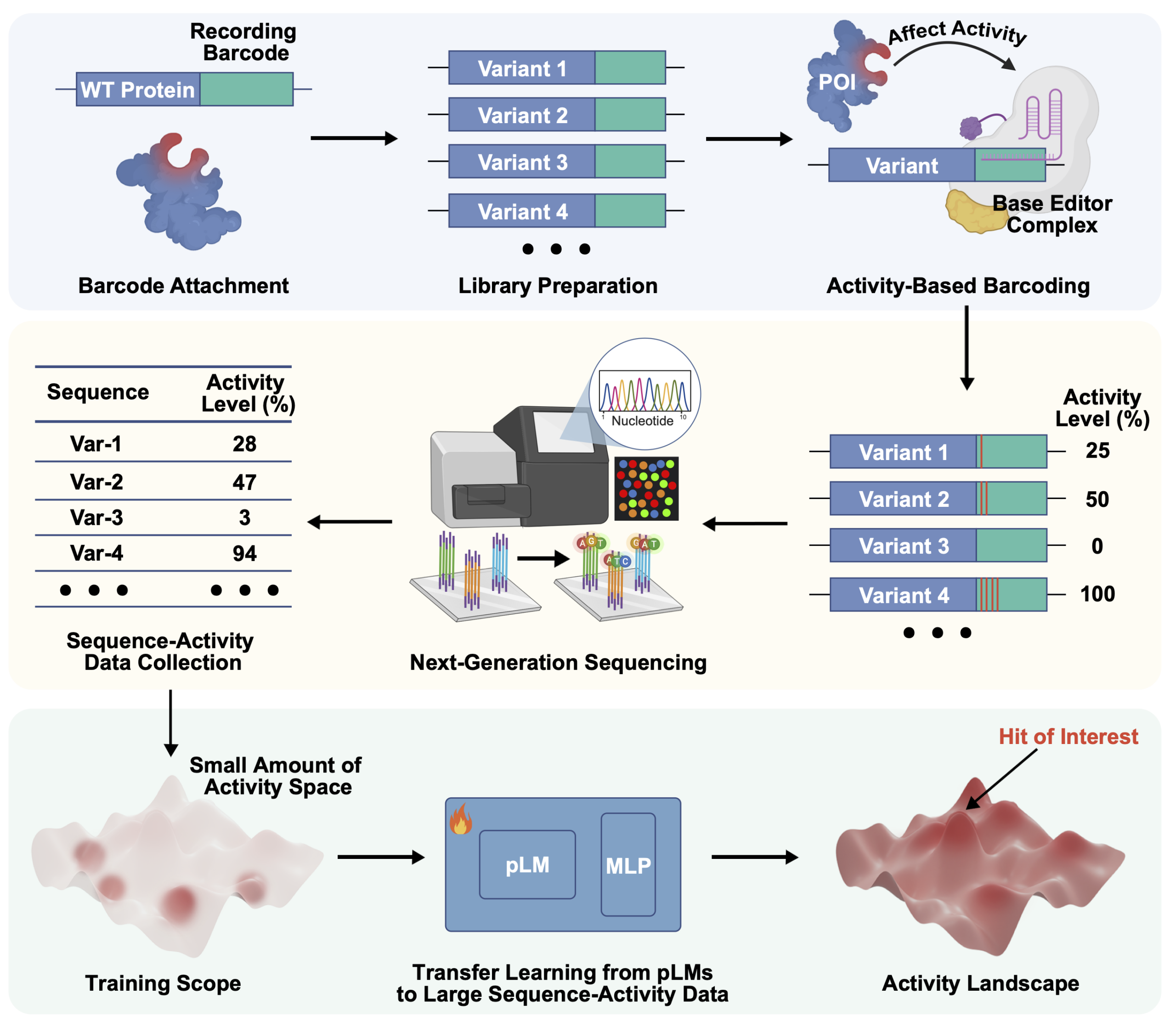
17. Sequence display: Generating large-scale sequence–activity datasets to advance protein evolution
Cheng, L.†; Zheng, X.†; Jiang, S.†; Hu, Y.; Liu, Y.; Yang, K.; Rui, J.; Ding, H.; Zhang, M.; Yuan, T.; Lu, Q.; Ye, H.; Li, C. L.; Guo, Y.; Tian, Z.; Qin, A.; Zhou, B.; Yang, K. K.; Huang, X.*; Xiao, H.* Nat. Biotech. 2026, published. [Paper Link] [Free text Link]
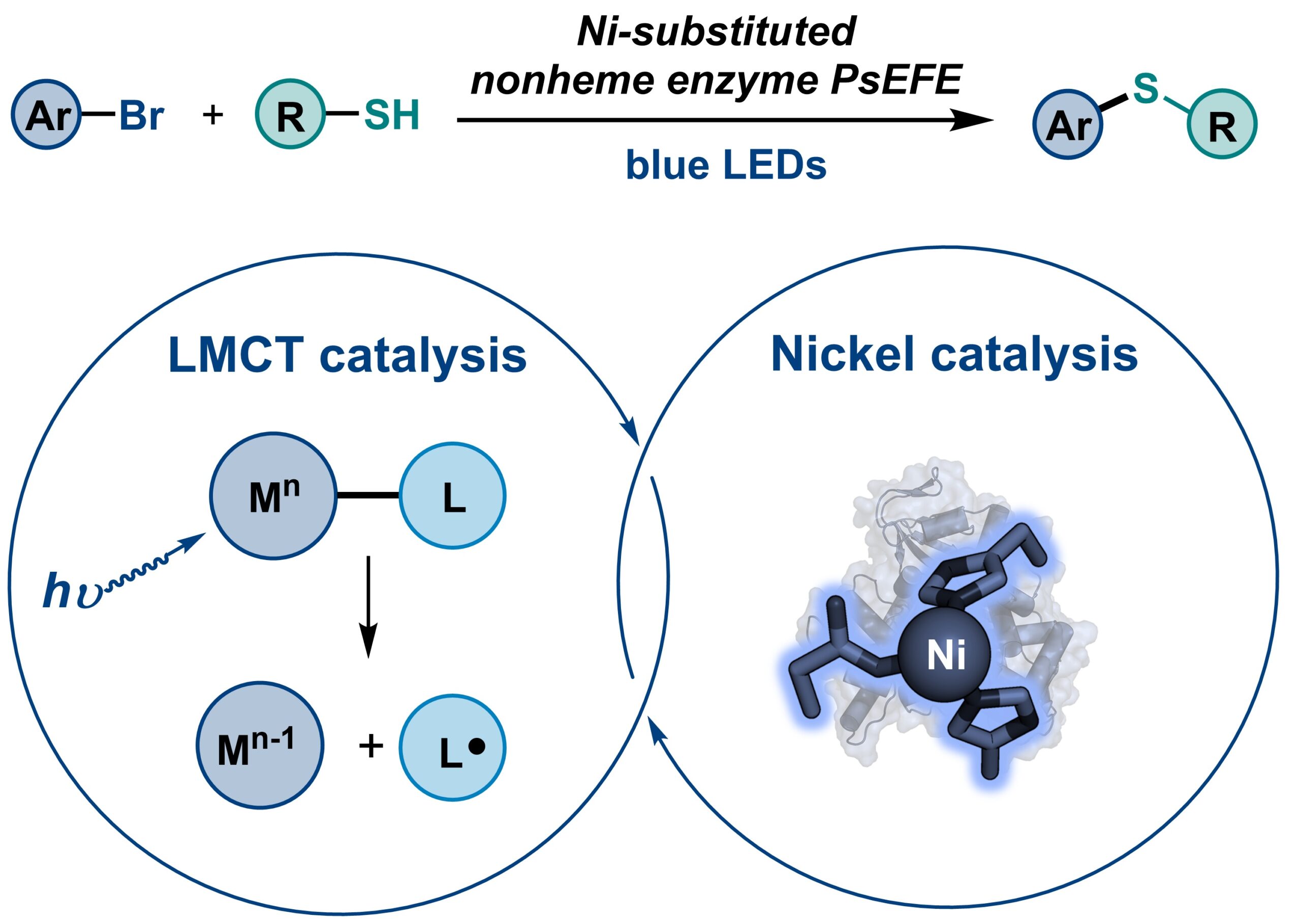
16. Engineering non-haem enzymes for nickel-catalyzed C(sp2)‒S coupling via ligand-to-metal charge transfer photocatalysis
Wang, X.†; Tian, X.†; Chen, X.†; Xu, G.; Paris, J.; Song, Y.; Su, Y.; Zhang, J. G.; Hong, X.; Garcia-Borràs, M.; Bragg, A. E.*; Guo, Y.*; Yang, Y-F.*; Huang, X.* Nat. Synth. 2026, published. [Paper Link] [Free text Link]
Highlighted in Nature Synthesis: “Light-driven metalloenzymatic C(sp²)–S cross-coupling”
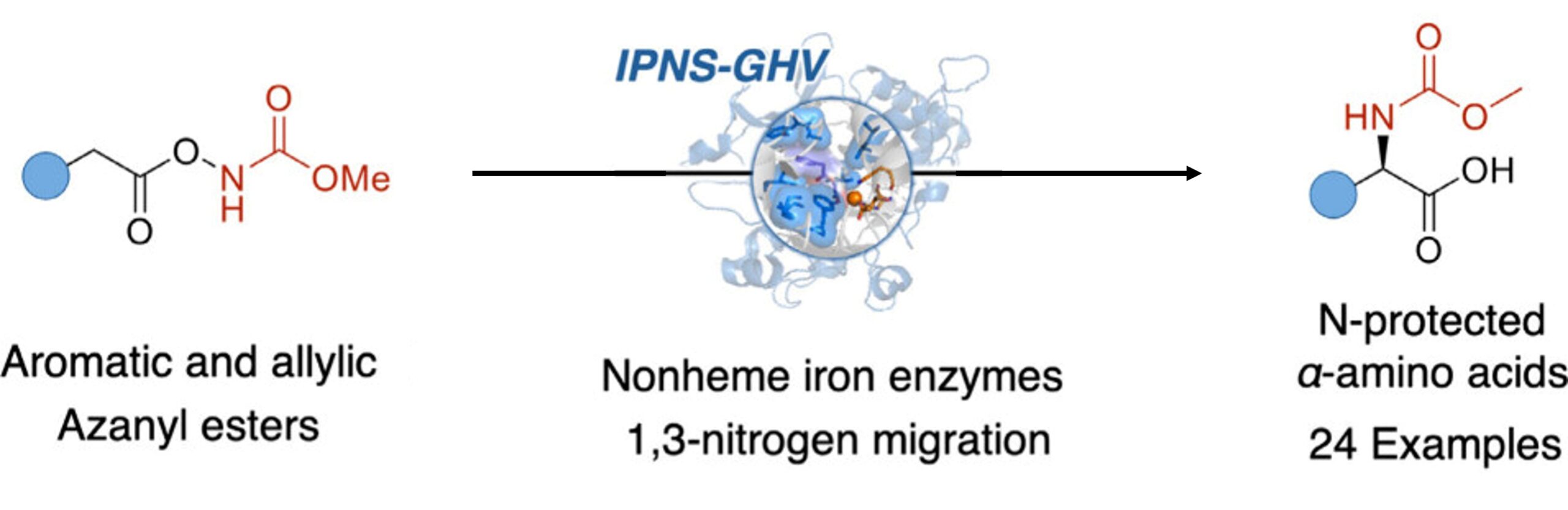
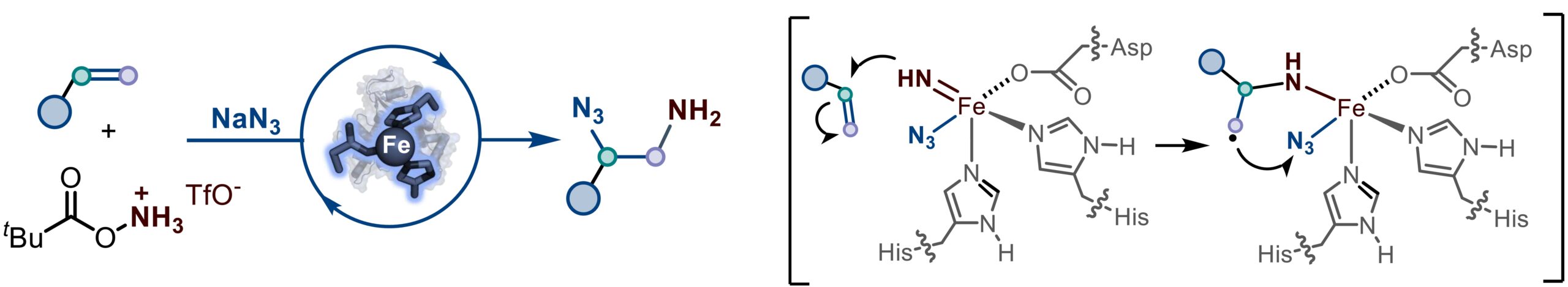
15. Biocatalytic synthesis of N-protected-amino acids through 1,3-nitrogen migration by nonheme iron enzymes
Yuan, T.†; Zhang, M.†; Chen, L.; Zhang, X.; Jiang, S.; Huang, X.*; Xiao, H.* J. Am. Chem. Soc. 2025, 147, 44041–44047. [Paper Link]
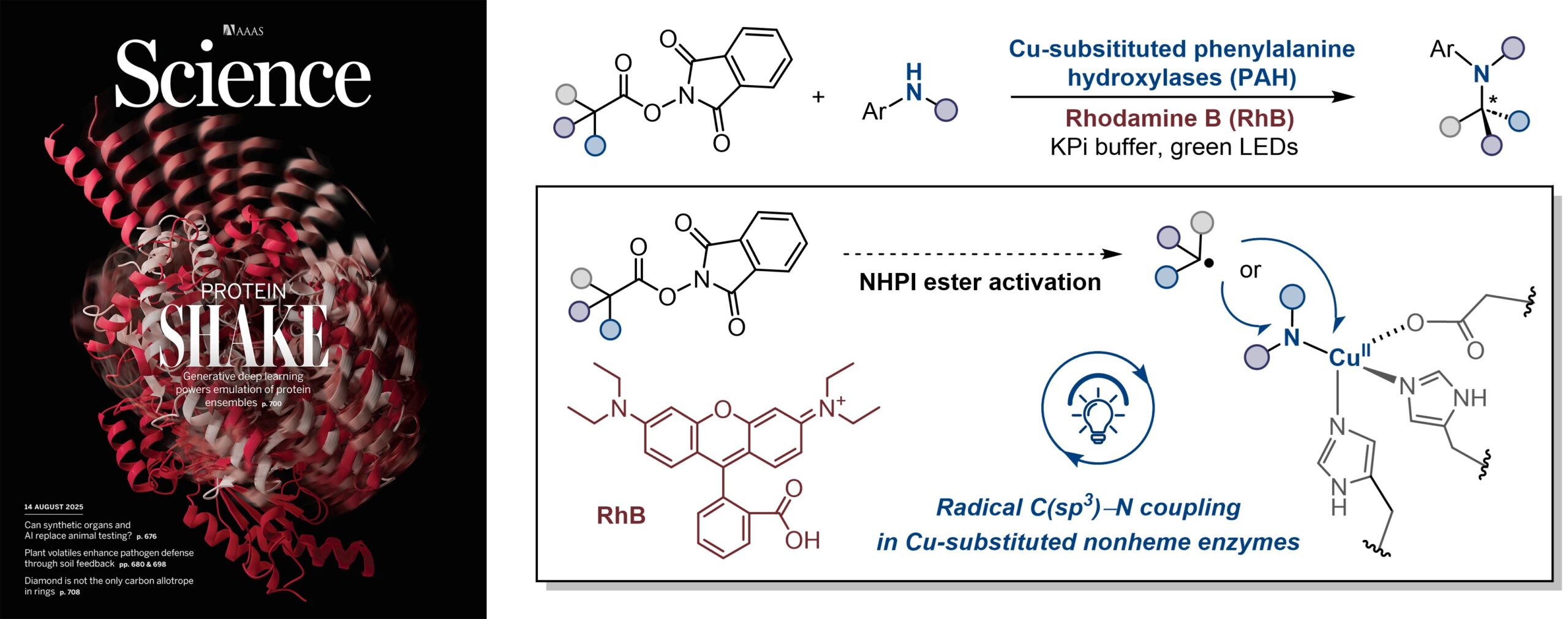
14. Enantioconvergent benzylic C(sp3)‒N coupling with a copper-substituted nonheme enzyme
Shen, X.; Chen, X.; Xiao, Y.; Zhang, J. G.; Brown, J. B.; Ji, X.; Rui, J.; Garcia-Borràs, M.; Rao, Y.*; Yang, Y-F.*; Huang, X.* Science 2025, 389, 741–746. [Paper Link]
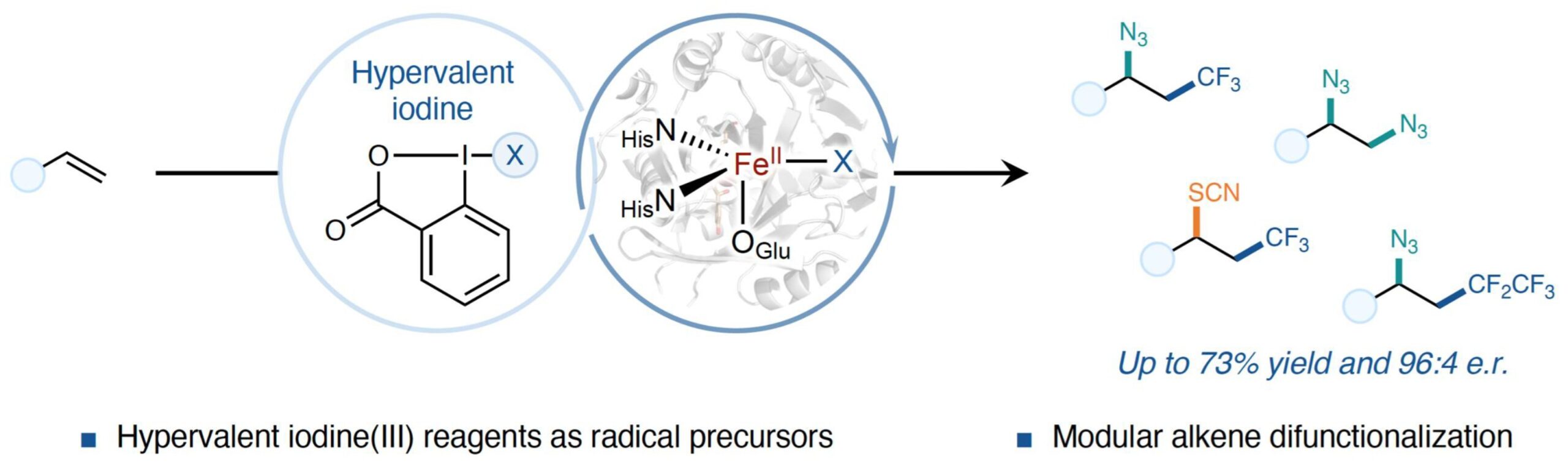
13. Biocatalytic olefin difunctionalization for synthesis of chiral 2-azidoamines using nonheme iron enzymes
Huls, A. J.†; Soler, J.†; Su, Y.; Yang, Y-F.; Garcia-Borràs, M.*; Huang, X.* Angew. Chem. Int. Ed. 2025, 64, e202423403. [Paper Link]
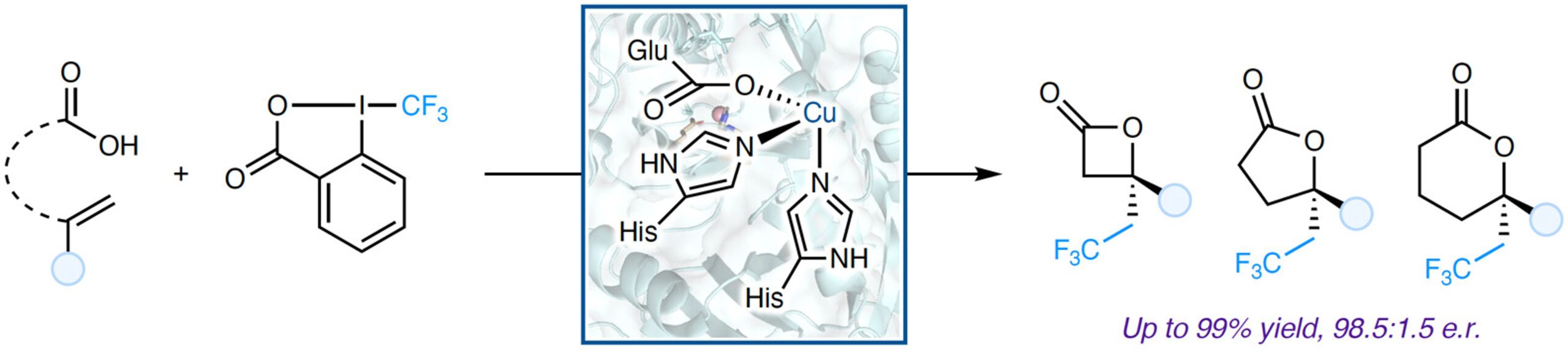
12. Directed evolution of copper-substituted nonheme enzymes for enantioselective alkene oxytrifluoromethylation
Zhang, J.; Huang, X.* J. Am. Chem. Soc. 2025, 147, 29624–29630. [Paper Link]
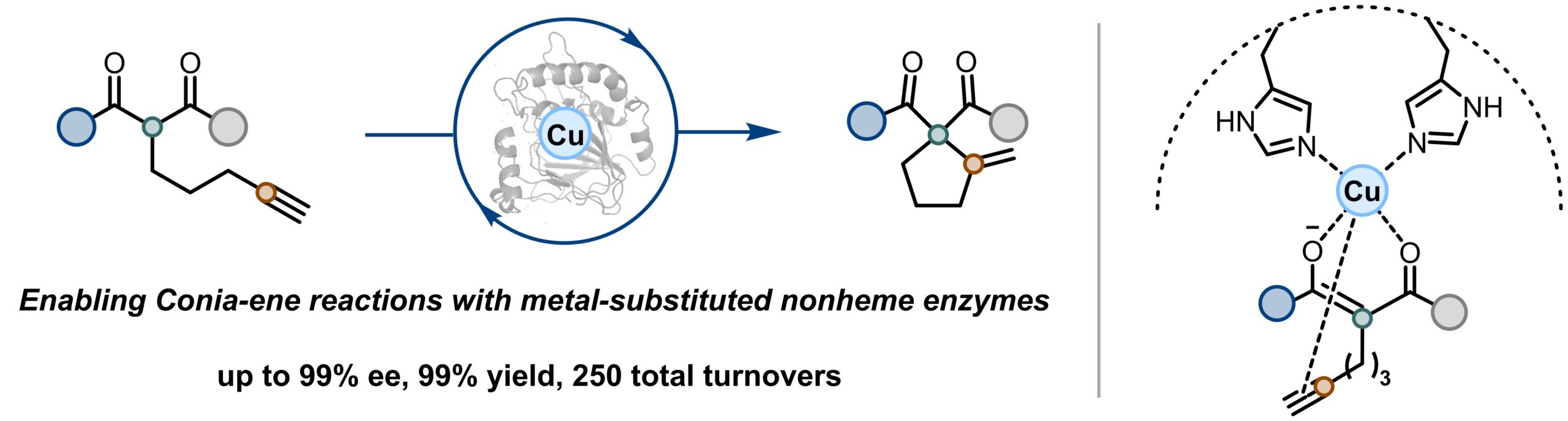
11. Unlocking Lewis acid catalysis in non-haem enzymes for an abiotic ene reaction
Mu, X.†; Ji, X.†; Chen, X.†; Wu, H.; Rui, J.; Hong, X.; Worth, M. M.; Reitz, A. D.; Goldberg, L. T. M.; Garcia-Borràs, M.; mIchel, S. L. J.; Yang, Y-F.*; Huang, X.* Nat. Catal. 2025, 8, 635–644. [Paper Link] [Free text Link]
Highlighted in Nature Catalysis: “A copper enzyme for Lewis acid biocatalysis”
10. Reprogramming nonheme iron enzymes for abiotic alkene trifluoromethylazidation
Zhang, J. G.; Huang, X.* Methods Enzymol. 2025, 720, 219–237. (invited contribution to the “Artificial Metalloproteins” issue) [Paper Link]
9. A metallophotoredox strategy for biocatalytic radical C–N3 and C–SCN bond formation
Rui, J.; Mu, X.; Huang, X.* Methods Enzymol. 2025, 721, 19–38. (invited contribution to the “Enzyme Reprogramming” issue) [Paper Link]
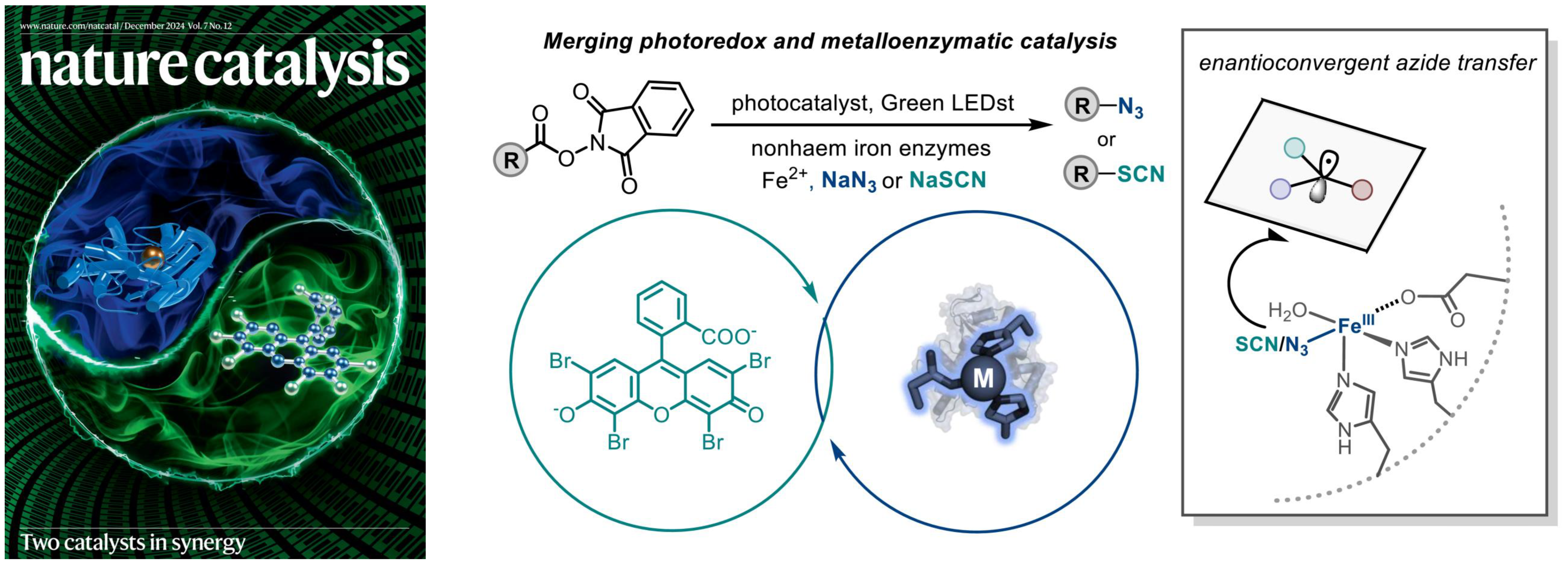
8. Merging photoredox with metalloenzymatic catalysis for enantioselective decarboxylative C(sp3)–N3 and C(sp3)–SCN bond formation
Rui, J.†; Mu, X.†; Soler, J.; Paris, J. C.; Guo, Y.*; Garcia-Borràs, M.*; Huang, X.* Nat. Catal. 2024, 7, 1394–1403. [Paper Link] [Free text Link]
Featured on the cover of Nature Catalysis (December 2024) [Link]
Further insights into the development of this work by our group can be found in this Nature Research Briefing [Link]
Highlighted in Synfacts: “Photoredox- and metalloenzyme-catalysis enables enantioselective radical azidations and thiocyanations”
7. Biocatalytic generation of trifluoromethyl radicals by nonheme iron enzymes for enantioselective alkene difunctionalization
Zhang, J. G.†; Huls, A. J.†; Palacios, P. M.; Guo, Y.*; Huang, X.* J. Am. Chem. Soc. 2024, 146, 34878–34886. [Paper Link] [ChemRxiv Link]
Highlighted in Synfacts: Enzymatic enantioselective radical trifluoromethyl azidation of styrenes”
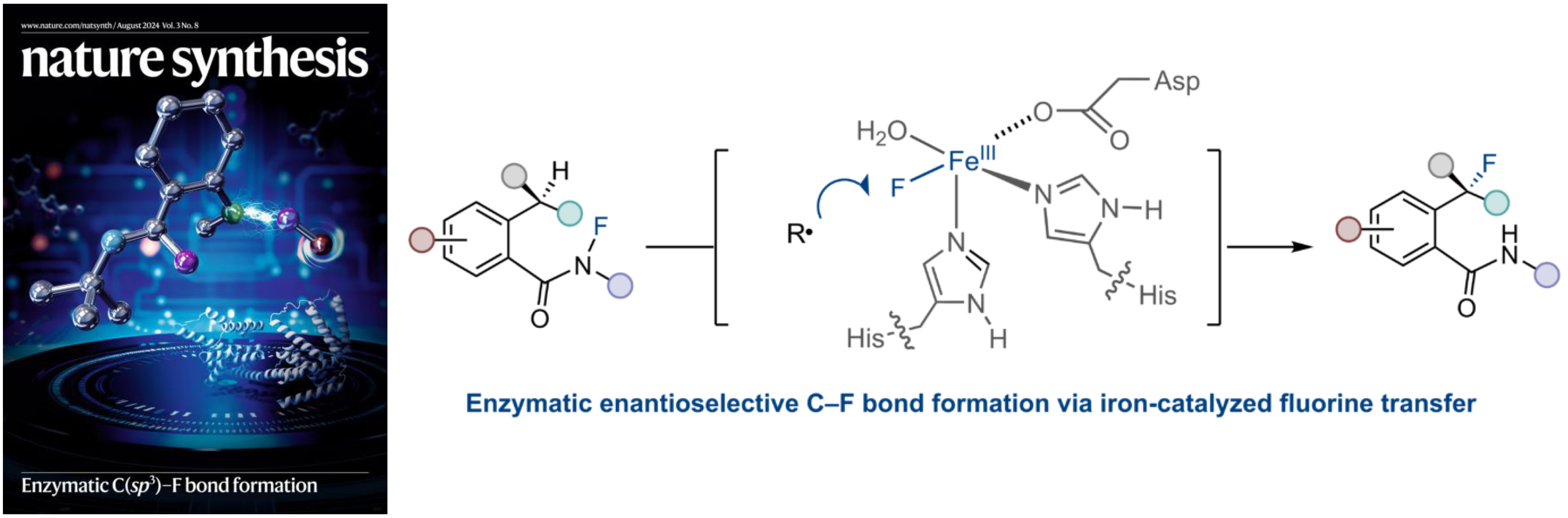
6. Engineering non-haem iron enzymes for enantioselective C(sp3)–F bond formation via radical fluorine transfer
Zhao, Q.†; Chen, Z.†; Soler, J.†; Chen, X; Rui, J.; Ji, N. T.; Yu, E. Q.; Yang, Y-F.*; Garcia-Borràs, M.*; Huang, X.* Nat. Synth. 2024, 3, 958–966. [Paper Link] [Free text Link]
Featured on the cover of Nature Synthesis (August 2024) [Link]
Highlighted in Nature Catalysis: “Enzymatic radical fluorine transfer”
Highlighted in Nature Synthesis: “A radical development for enzymatic fluorination“
5. Radical fluorine transfer catalysed by an engineered nonheme iron enzyme
Zhao, Q.; Chen, Z.; Soler, Rui, J.; Huang, X.* Methods Enzymol. 2024, 696, 231–247. (invited contribution to the “Fluorine Metabolism, Transport and Enzymatic Chemistry” issue) [Paper Link]
4. Radical-relay C(sp3)–H azidation catalyzed by an engineered nonheme iron enzyme
Zhao, Q.*; Rui, J.; Huang, X.* Methods Enzymol. 2024, 703, 195–213. (invited contribution to the “Mononuclear Non-heme Iron Dependent Enzymes” issue) [Paper Link]
3. Protoglobin-catalyzed formation of cis-trifluoromethyl-substituted cyclopropanes by carbene transfer
Schaus, L.†; Das, A.†; Knight, A. M.; Jimenez-Osés, G.; Houk, K. N.; Garcia-Borràs, M.*; Arnold, F. H.*; Huang, X.* Angew. Chem. Int. Ed. 2023, e202208936. [Paper Link] [Free text Link]
2. The many facets of green organometallic chemistry: A foreword
Huang, X.; Yang, Y. J. Organomet. Chem. 2022, 976, 122398. [Paper Link]
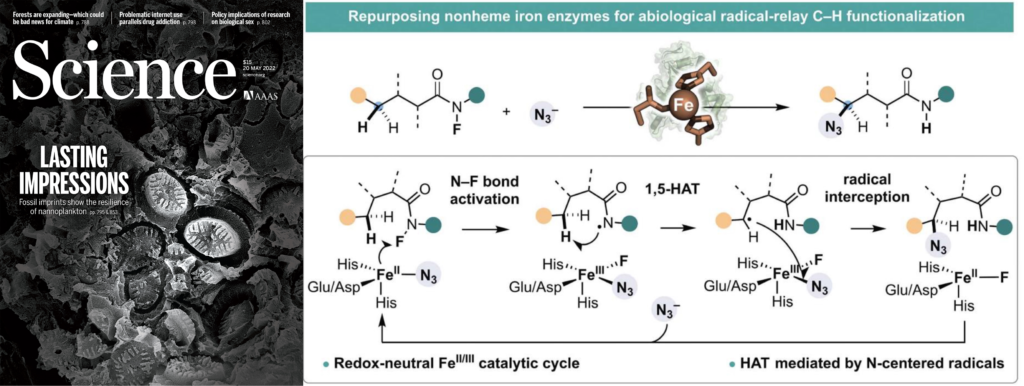
1. Directed evolution of nonheme iron enzymes to access abiological radical-relay C(sp3)−H azidation
Rui, J.†; Zhao, Q.†; Huls, A. J.†; Soler, J.; Paris, J. C.; Chen, Z.; Reshetnikov, V.; Yang, Y-F.; Guo, Y.*; Garcia-Borràs, M.*; Huang, X.* Science 2022, 376, 869-874. [Paper Link] [Free text Link]
Highlighted in Chem Catalysis: “A radical-relay approach to biocatalytic C–N bond formation”
Highlighted in Chem Catalysis: “Directed evolution of non-heme iron enzymes to access a non-natural radical-relay C(sp3)−H azidation”
Caltech
7. Enantiodivergent α-Amino C−H Fluoroalkylation Catalyzed by Engineered Cytochrome P450s
Zhang, J.†; Huang, X.†; Zhang, R. K.; Arnold, F. H.* J. Am. Chem. Soc. 2019, 141, 9798-9802. (†equal contribution) [Link]
6. A Biocatalytic Platform for Synthesis of Chiral α-Trifluoromethyl Organoborons
Huang, X.†; Garcia-Borràs, M.†; Miao, K.; Kan, S. B. J.; Zutshi, A.; Houk, K. N.*; Arnold, F. H.* ACS Cent. Sci. 2019, 5, 270-276. (†equal contribution) [Link]
[Featured in ACS Central Science and Nature Catalysis]
5. Engineered Cytochrome c-Catalyzed Lactone-Carbene B–H Insertion
Chen, K.; Huang, X.; Zhang, S. Q.; Zhou, A. Z.; Kan, S. B. J.; Hong, X.*; Arnold, F. H.* Synlett. 2019, 30, 378-382. [Link]
4. Enzymatic Assembly of Carbon–Carbon Bonds via Iron-Catalysed sp3 C–H Functionalization
Zhang, R. K.; Chen, K.; Huang, X.; Wohlschlager, L.; Renata, H.; Arnold, F. H.* Nature 2019, 565, 67-72. [Link]
[Selected as the Nature cover article, featured in Synfacts]
3. Selective C−H Bond Functionalization with Engineered Heme Proteins: New tools to Generate Complexity
Zhang, R. K.; Huang, X.; Arnold, F. H.* Curr. Opin. Chem. Biol. 2019, 49, 67-75. [Link]
2. Enzymatic Construction of Highly Strained Carbocycles
Chen, K.; Huang, X.; Kan, S. B. J.; Zhang, R. K.; Arnold, F. H.* Science 2018, 360, 71-75. [Link]
[Featured in Caltech News, Phys.org, myScience, and EurekAlert]
1. Genetically Programmed Chiral Organoborane Synthesis
Kan, S. B. J.†; Huang X.†; Gumulya, Y.; Chen, K.; Arnold, F. H.* Nature 2017, 552, 132-136. (†equal contribution) [Link]
[Featured in Caltech News, GEN, myScience, Forbes, ScienceNewsline, EurekAlert, Phys.org]
Princeton
12. Site-Selective 18F Fluorination of Unactivated C−H Bonds Mediated by a Manganese Porphyrin
Liu, W.; Huang, X.; Placzek, M. S.; Krska, S. W.; McQuade, P.; Hooker, J. M.*; Groves, J. T.* Chem. Sci. 2018, 9, 1168-1172. [Link]
11. Oxygen Activation and Radical Transformations in Heme Proteins
Huang, X.; Groves, J. T.* Chem. Rev. 2018, 118, 2491-2553. [Link]
10. Alkyl Isocyanates via Manganese-Catalyzed C–H Activation for the Preparation of Substituted Ureas
Huang, X.†; Zhuang, T.†; Kates, P. A.; Gao, X.; Chen, X.; Groves, J. T.* J. Am. Chem. Soc. 2017, 139, 15407–15413. (†equal contribution) [Link]
9. The Enigmatic P450 Decarboxylase OleT Is Capable of, but Evolved to Frustrate, Oxygen Rebound Chemistry
Hsieh, C. H.; Huang, X.; Amaya, J. A.; Rutland, C. D.; Keys, C. L.; Groves, J. T.; Austin, R. N.; Makris, T. M. Biochemistry 2017, 56, 3347–3357. [Link]
8. Beyond Ferryl-Mediated Hydroxylation: 40 Years of the Rebound Mechanism and C−H Activation
Huang, X.; Groves, J. T.* J. Biol. Inorg. Chem. 2016, 1–23. [Link]
7. Taming Azide Radicals for Catalytic C−H Azidation
Huang, X.; Groves, J. T.* , ACS Catal. 2016, 6, 751–759. [Link]
6. Manganese-Catalyzed Late-Stage Aliphatic C−H Azidation
Huang, X.; Bergsten, T. M.; Groves, J. T.* J. Am. Chem. Soc. 2015, 137, 5300–5303. [Link]
5. Targeted Fluorination with the Fluoride Ion by Manganese-Catalyzed Decarboxylation
Huang, X.; Liu, W.; Hooker, J. M.; Groves, J. T.* Angew. Chem. Int. Ed. 2015, 54, 5241–5245. [Link]
[Selected as a “Hot Paper” by Angewandte Chemie.]
4. Late Stage Benzylic C−H Fluorination with [18F]Fluoride for PET Imaging
Huang, X.†; Liu, W.†; Ren, H.; Neelamegam, R.; Hooker, J. M.*; Groves, J. T.* J. Am. Chem. Soc. 2014, 136, 6842–6845. (†equal contribution) [Link]
[Highlighted in C&EN News, Angew. Chem. Int. Ed. and Chem. Sci.]
3. Oxidative Aliphatic C−H Fluorination with Manganese Catalysts and Fluoride Ion
Liu, W.; Huang, X.; Groves, J. T.* Nat. Protoc. 2013, 8, 2348–2354. [Link]
2. Oxidative Aliphatic C−H Fluorination with Fluoride Ion Catalyzed by a Manganese Porphyrin
Liu, W.; Huang, X.; Cheng, M.; Nielsen, R. J.; Goddard, W. A. III; Groves, J. T.* Science 2012, 337, 1322–1325. [Link]
[Highlighted in Nature, C&EN News, Chemistry World, Princeton University home page]
1. Parallel and Competitive Pathways for Substrate Desaturation, Hydroxylation and Radical Rearrangement by the Non-heme Diiron Hydroxylase AlkB
Cooper, H. L. R.; Mishra, G.; Huang, X.; Pender-Cudlip, M.; Austin, R. N.; Shanklin, J.; Groves, J. T.* J. Am. Chem. Soc. 2012, 134, 20365–20375. [Link]
USTC
4. Hydride Dissociation Energies of Six-Membered Heterocyclic Organic Hydrides Predicted by ONIOM-G4 Method
Shi, J.*; Huang, X.; Wang, H. J.; Fu, Y. J. Chem. Inf. Model. 2012, 52, 63–75. [Link]
3. A Theoretical Study on C−COOH Homolytic Bond Dissociation Enthalpies
Shi, J.*; Huang, X.; Wang, J. P.; Li, R. J. Phys. Chem. A 2010, 114, 6263–6272. [Link]
2. Theoretical Study on Acidities of (S)-Proline Amide Derivatives in DMSO and Its Implications for Organocatalysis
Huang, X.; Wang, H. J.; Shi, J.* J. Phys. Chem. A 2010, 114, 1068–1081. [Link]
1. Theoretical Study of One-Electron Redox Potentials of Some NADH Model Compounds
Wang, H-J.; Huang, X.; Shen, R.; Fu, Y.*; Rui, L. Chin. J. Chem. 2010, 28, 72–80. [Link]